
Fig. 1. First salt, then slush and a plough, and finally a snow blower – often have detrimental effects on nature in an area. Photo: Tiefbauamt Graubünden
Winter road treatment is a public service and is intended to keep the users of public paths and roads safe. It includes making efforts to minimise slipping hazards when there is snow, frost or ice. Should improper maintenance result in an accident, the owner of the road is held liable (Swiss Code of Obligations, Art. 58).
As traffic volumes grow and the speed of travel increases, the pressure on public infrastructure is mounting and needs are becoming harder and harder to meet. An average of 300,000 to 350,000 t of de-icing salts – mainly sodium chloride (NaCl) – was spread on Switzerland's roads each winter during the last few years. Depending on the place and the weather conditions, each m2 of road surface received a total of between 100 and over 1,000 g of NaCl each winter.
Direct damage
In Switzerland, direct damage to concrete surfaces and to concrete and steel structures caused by de-icing salt totals 5 to 6 billion CHF a year, while corrosion damage to vehicles comes in at several hundred million CHF each year. The direct effects on water cannot be quantified. For a fully accurate picture of the costs of the damage caused by de-icing salt, we would have to take into account the expenses associated with road drainage and pipeline flushing, the cost of special precautions taken to ensure the smooth operation of sewage treatment plants and environmental effects.
Effects on trees
For instance, a comparison conducted for the City of Basel showed that trees that have sustained salt damage generally only live 60% as long as undamaged trees. However, the average annual maintenance cost over the tree lifetime is no lower for undamaged than for damaged trees: Decisive is the increased expenditure for maintenance activities like pruning and hazard elimination. Nevertheless, these calculations do not take into account the loss of the ecological performance of sick and diseased trees or the loss of their intangible, cultural value and their value for urban planning and design. Offsetting the ecological loss of these older trees alone would require hundreds of new trees to be planted and maintained, in addition to maintaining the trees damaged by salt. This would significantly increase costs.

Fig. 2. Salt damage after several years of contamination through the salt cycle. Photo: City of Basel Parks Department
How de-icing salt gets into the environment
When dry de-icing salt is spread on roads – a method still in common use – only some of the salt remains on the road surface. Large amounts of salt are displaced by the wind and by passing vehicles. This salt then ends up on nearby surfaces or on trees and shrubs, either as salt dust or as a suspension in the damp air (aerosol).
Salt that has dissolved in snowmelt is channelled through drains into surface water, where it is diluted, or travels through pipes to sewage treatment plants. Vehicles also spray this salt solution to the sides of the road, where it lands on the ground vegetation and trees at the roadside.
Ploughed snow deposited along the sides of roads has particularly harmful effects on trees and vegetation. The salt contained in ploughed snow lands directly on exposed leaves, branches and bark, or seeps into the ground when the snow melts.
Snow blowers are used purely to remove large quantities of snow. "Out of sight, out of mind" is usually the motto extended to the salt-laden ploughed snow. This is mainly carried out in residential areas with hedges and trees, but is also fairly common along narrow mountain roads outside of built-up areas.
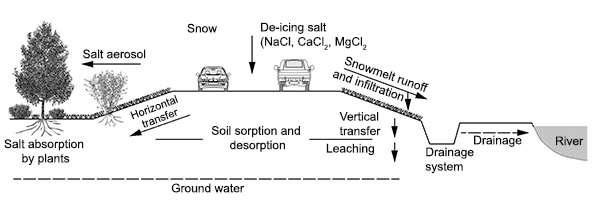
Fig. 3. How de-icing salt end up in the environment: Mechanically spread de-icing salt get into the environment in a range of ways and damages plants, soil and water. Click to enlarge. Salt absorption by plants. Source: from Brod, H.G., 1993
Effects of de-icing salt on plants and soil

Fig. 4. This situation was probably caused by de-icing salt. Photo: Swiss Forest Protection

Fig. 5. Leaves of a horse chestnut tree that has been damaged by de-icing salt. Photo: Swiss Forest Protection
When salt comes into direct contact with plants, it penetrates tissues and affects plant metabolism. The tissues dry out and burn, resulting in browned needles and leaves. However, these signs of damage are not visible until spring and summer.
When salt enters the soil, it changes the soil fabric. Sodium and chloride ions are then carried to plants in an indirect way through the soil solution. They are then absorbed by the plants' roots and disrupt the balance of nutrients within plants. This imbalance mainly interferes with metabolic processes and destroys cell structures, but it can also cause drought stress. The most visible signs of damage to the entire system are scorched leaf edges and leaf necrosis. The highest salt concentrations in plants are found at the edges of leaves.
Vicious salt cycle
When an area is heavily contaminated, salt ions are everywhere:
- they run off of needles and leaves;
- they are leached from leaf tissues;
- they enter the soil when leaves fall prematurely;
- they are stored in plant storage organs (buds, branches, trunk, roots);
- they enter the nutrient solution when the autumn leaves decompose;
- they are fed directly into the soil in salt dust, salt spray, ploughed snow and snowmelt;
- they are stored in the soil solution;
- they are adsorbed onto soil particles;
- they are absorbed by plant roots;
- they are transported through plant conductive vessels to the needles and leaves.
Because salt is absorbed into the ecosystem (substrate, understory vegetation, plant) as a solution and is only partly removed – e.g. through soil leaching – most of it remains in the system and salt levels increase as new salt is added each winter. The trees can never recover fully and are subject to continuous stress.
The seasonal addition of new salt results in a vicious cycle involving the tree crown, the soil and the roots. Due to this salt cycle (storage in buds and branches in winter, plus transfer of Na+ and Cl– from the leaves to the roots and back to the plant), salt levels in trees can climb constantly over the years and eventually reach lethal concentrations.
One highly visible example is the dying trees seen in exposed places where there is intensive road treatment in winter, e.g. at bus stops or pedestrian crossings or along shady sections of mountain roads.
Salt tolerance of tree species is always relative
Not all plants that occur in nature are glycophytes (non-salt-tolerant plants). Some are halophytes (salt-tolerant plants) and cope better with high salt concentrations. However, all the trees and shrubs found in urban areas and forests are glycophytes. They are not completely unable to tolerate salt, but tolerance varies among species and there are even genetic differences within species. Because trees are so long-lived, it would be difficult to carry out selective breeding using their seeds. Besides, each individual seed has a distinct combination of genes. Conversely, experiments with vegetative propagation, e.g. using cuttings, have demonstrated some degree of success.
Plants – or at least glycophytes – cannot be salt resistant. However, their degree of salt tolerance is determined by other parameters, including their constitution, their surroundings and other influencing factors.
It is difficult to quantify how much of the salt spread on roads ends up in each of the different 'destinations' (plants, road drainage, soil and so on). Further, it is not possible to draw general conclusions about the salt concentrations measured in the soil and in leaves/needles. Even leaves that show no outward signs of damage may contain high levels of salt. Moreover, salt concentrations reveal little about how much salt was actually spread the previous winter.
Measures to prevent salt damage
Fewer and fewer tree species are suitable for planting in urban areas. Factors such as biodiversity, location, maintenance, appearance and effect on the human population all play a part when it comes to selecting trees. Other important considerations include vulnerability to pathogens, adaptability to climate change, drought stress tolerance and sensitivity to air pollutants. Invasive species must not be used under any circumstances. The range of options is even narrower when it comes to planting new trees in a forest, as near-natural forestry should involve as few risks as possible. The only tree species that should be planted in such cases are those that are native to or, at the very least, appropriate for the location.
As shown by a comprehensive study of the literature on this subject, there is ultimately considerable potential for avoiding the damage caused by spreading de-icing salt for winter road treatment. The primary means of reducing this damage is to use the correct dose of salt. Excess salt can be counterproductive for road users, too, and has an impact on indirect costs. Spreading salt is, in principle, pointless at times of heavy snowfall, as the resulting slush makes the road surface far more slippery. In these situations, snow must be cleared by mechanical means instead. All of this applies not only to sodium chloride (NaCl), the most commonly used de-icing salt, but also to other salts like magnesium chloride (MgCl2) and calcium chloride (CaCl2).
Key ways to reduce the amount of salt spread:
• A winter road treatment plan that takes into account different road types;
• Optimisation of salt-spreading techniques and adjustment of salt doses to match the situation, combined with use of appropriate equipment, staff training and briefing, and spreading of salt in pre-wetted or brine form;
• Preventive salt-spreading at the right time using the right technique;
• Selection of alternative road grits for roads with lower traffic volumes, cycle paths, footpaths, etc., or even partial suspension of snow-clearing providing appropriate signage is posted;
• Reorientation of the award of winter road treatment contracts to private companies, with award criteria based not on the total amount of salt spread, but on overall readiness to operate according to the winter road treatment plan while limiting the amount of salt spread.
Translation : TTN Translation Network
